Given The Equation Representing A System At Equilibrium
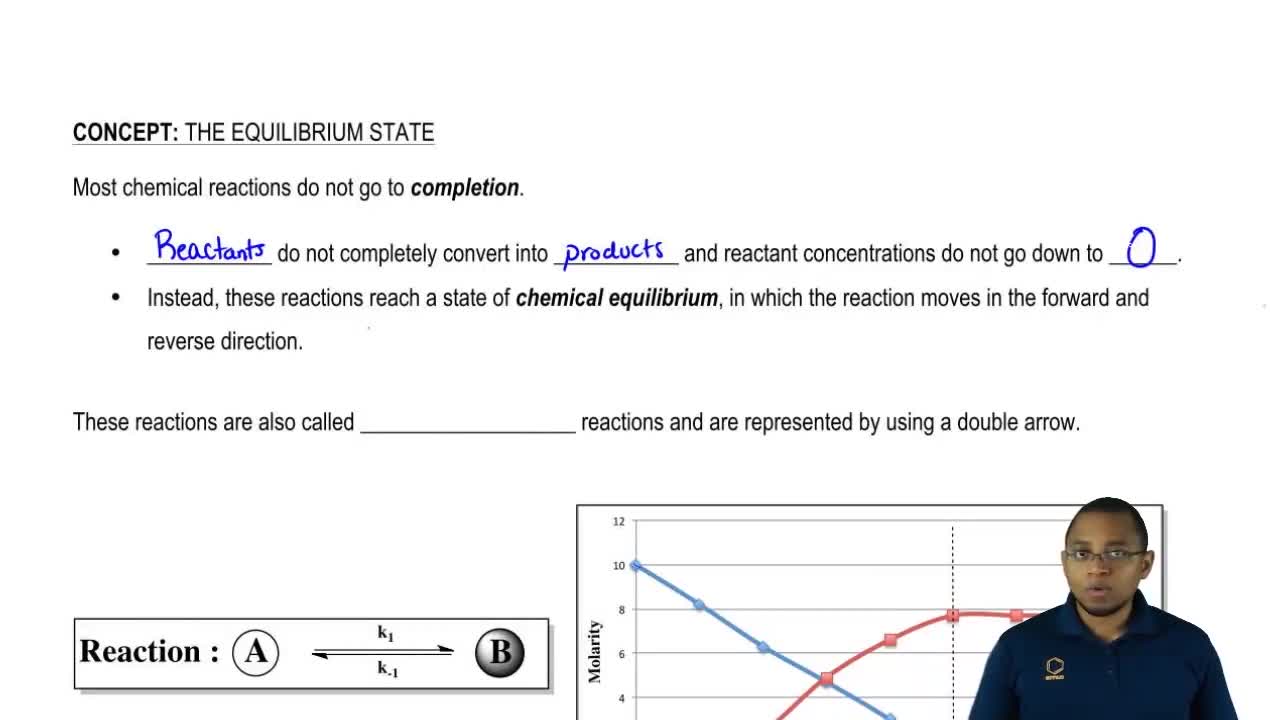
Given the equation representing a system at equilibrium. Get the answers you need now. What is an equilibrium equation. N2O4g 2 NO2 g shows that the concentration of N2O4 g and the concentration of NO2 g is constant.
PCl5g energy PCl3g Cl2g Which change will cause the equilibrium to. PCl 5 g PCl 3 g Cl 2 g. N2g 3H2g 2NH3g What occurs when the concentration of H2g is increased.
20Given the equation representing a reaction at equilibrium. Correct answer - Given the equation representing a system at equilibrium. In the static case the equilibrium equation is.
Given the equation representing a system at equilibrium. A system at equilibrium as described by this equation. A activation energy decreases B activation energy increases Cfrequency of effective collisions decreases D frequency of effective collisions increases 21Given the balanced equation representing a.
This is true because the rate at which the forward reaction is occurring that is N2O4 giving NO2 is the same as the rate at which the reverse reaction is occurring. 12Given the equation representing a system at equilibrium. N2g 3H2g 2NH3g energy Which changes occur when the temperature of this system is decreased.
1 increasing pressure 2 incr. Adecreasing the concentration of HIg Bdecreasing the temperature Cincreasing the concentration of I2g Dincreasing the pressure 13Given the equation representing a reaction at equilibrium. Given the equation representing a system at equilibrium.
623 where K is the stiffness matrix of the system u is the vector with the nodal displacements and F represents the external forces Fig. Given the equation representing a system at equilibrium.
PCl5g energy PCl3g Cl2g Which change will cause the equilibrium to.
Get the answers you need now. The concentration of N2O4g and the concentration of NO2g must be constant. Correct answer to the question Given the equation representing a system at equilibrium. Correct answer - Given the equation representing a system at equilibrium. 2HI g H2 g I2 g energy. In the static case the equilibrium equation is. Get the answers you need now. 20Given the equation representing a reaction at equilibrium. 1 increasing pressure 2 incr.
N2g 3H2g 2NH3g What occurs when the concentration of H2g is increased. Correct answer to the question Given the equation representing a system at equilibrium. Given the equation representing a system at equilibrium in a sealed rigid container. H2g I2g heat 2HIg. In the static case the equilibrium equation is. 1 increasing pressure 2 incr. PCl5g energy PCl3g Cl2g Which change will cause the equilibrium to shift to the right.


























Post a Comment for "Given The Equation Representing A System At Equilibrium"